As neurophysiologists, we require the use of some rather sophisticated electronic equipment to amplify and measure neural signals in the brain. Complex systems have been developed to efficiently record 10s to 100s of channels of neural data from increasingly small sensors implanted in the brain. That being said, sometimes the equipment is complicated… for basically no reason. At best, we might chalk this up to engineering laziness, at worst, a thinly veiled attempt to prevent customers from interfacing systems from different manufacturers. In this case, I think a manufacturer tricked even themselves.
The Silicon Probe
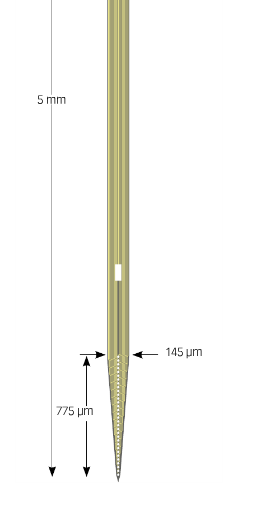
The A1x32-5mm-25-177 has 32 Channels on board
One type of hardware I use frequently is the microelecrode. This is a device that measures tiny voltage changes that happen outside cells (neurons) in the brain. These voltages reflect the activity of cells in the brain coding sensory and behavioral information. One particularly small and efficient microelectrode is the multi-channel silicon probe. These electrodes are thinner than a human hair but fit up to 64 conductive electrode pads on a single shank. The more channels of neural data you have, the more activity (cells) you can measure at once, allowing more complex and efficient data collection. In addition, the smaller the electrode, the less damage done to the brain tissue while measuring neural signals. Due to these advantages and more, I started using 32-channel silicon probes from NeuroNexus in 2014.
Datasheet Blues
This is not going to be a highly technical post. The basic gist is that at the end of the day I just need to know which signals are recorded from which sites on the electrode (the little white dots at the triangular tip of the silicon shank).

The A32-Z32 adapter
Unfortunately, the sites are not numbered 1,2,3,4… (etc) from one end, like you might expect. In fact from the top, the sites of the A1x32 probe are numbered 17,16,18,15,19 … etc. While at first annoying, this is ultimately a minor inconvenience: the manufacturers provide detailed datasheets describing how the electrode pads are numbered and I remap the sites accordingly.
One added wrinkle, however, is that I use an adapter to go from the silicon probe to the amplifier and data acquisition system. In this case, I bought the NeuroNexus A32-Z32 connector ($200) to go from the silicon probe to ZifClip headstage connectors that we already had in our lab. Of course, both the NeuroNexus A32-Z32 connector and the TDT system have their own arbitrary channel numbering systems. This leaves me with a series of four (!) channel re-mappings:
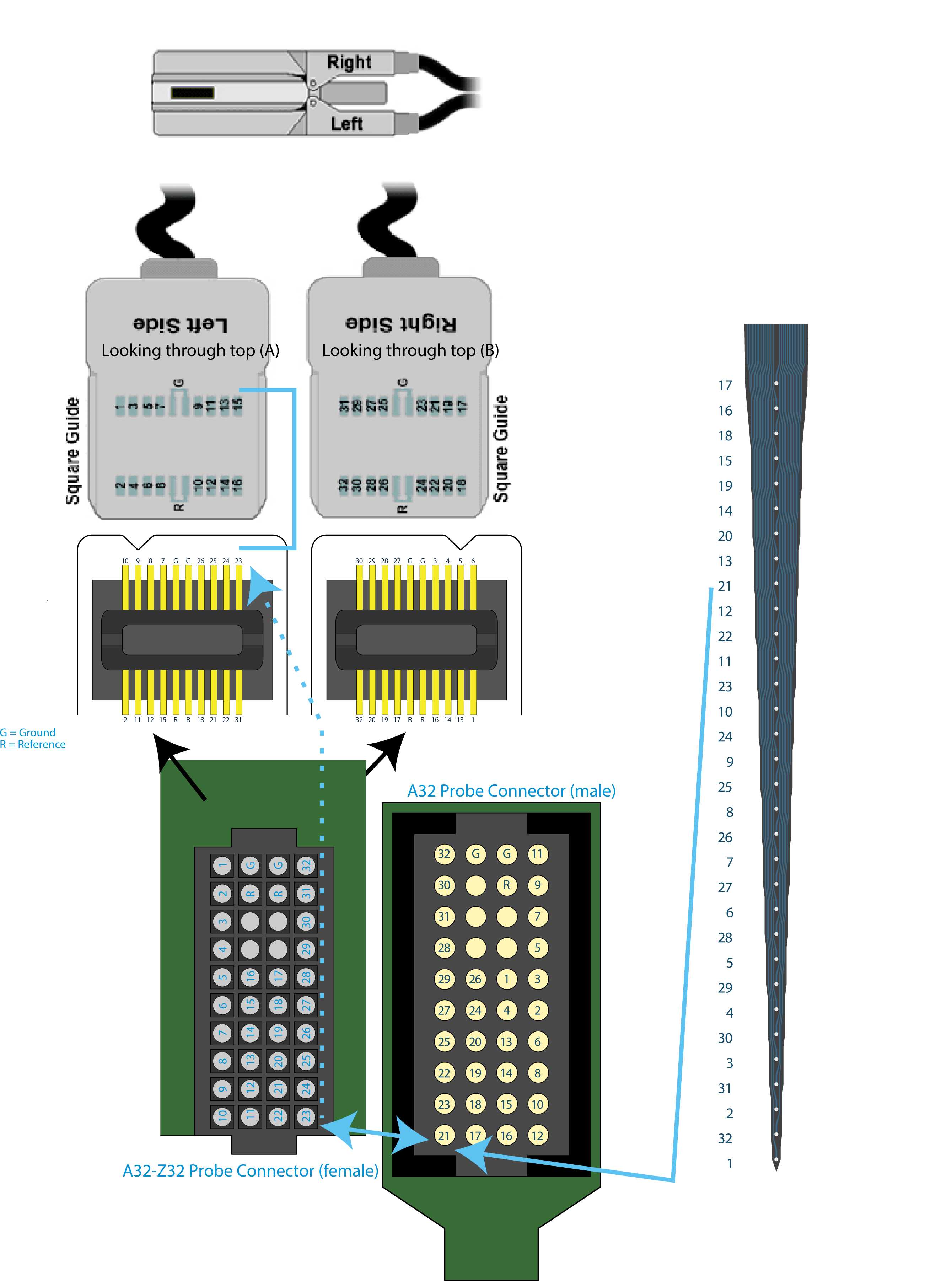
A1x32-to-A32-Z32-to-ZiffClip Channel Numbering. Note: This was “correct” up until very recently. (See below for a correct mapping)
For example, the channel I would call #9 if I counted from the top of the shank maps from 9(Counting) -> 21(NeuroNexus) -> 23 (A32-Z32, also NeuroNexus) -> 15 (ZifClip TDT). This is sort of insane, but whatever. As long as it works, it is solved: each manufacturer provides datasheets that allow me to map where the signal goes from one device to the next. Good. Done.
But… actually, not done. Not done at all.
Don’t Trust the Strange Data
I started to notice something strange in my neural recordings:
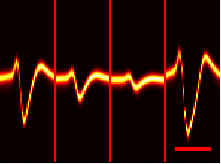
Four “adjacent” sites near one cell with large amplitude jumps from channel-to-channel
Because we amplify the signals of individual cells in the brain, the amplitude of the signal should not “jump” channel-to-channel like the example above. Instead, it should vary continuously across adjacent channels, with a peak signal on one channel:
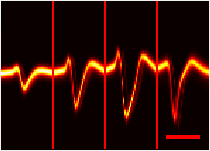
Four adjacent channels with smoothly varying signal amplitude.
I started to notice pretty consistent issues with a few of the channels. My first thought was that I messed up the tedious mapping shown above. So I double-, triple- and quadruple- checked the datasheets. I started feeling a bit crazy because I did not see anything wrong. I next decided to manually check the circuitry of the A32-Z32 adapter with a multimeter, which involved checking the resistance of all 32 channels on the A32 side of the adapter to all 32 channels on the Z32 (the low-resistance “beep” setting on multimeters is invaluable). If that doesn’t seem tedious enough on it’s own, consider that I needed to do the entire thing under a microscope because the adapter is about 1 x 2cm.
Number Me This
Low and behold, four channels did not match the datasheet. The way they were switched affected a large swath of the probe. I was still sort of incredulous that this was possible, so I decided to e-mail NeuroNexus to see if I had an outdated datasheet for the A32-Z32. They replied:
“This adaptor design hasn’t changed since its first build [2011], so this is the only [datasheet] we have. We will be contacting the other customers who have purchased this item to notify them of the error. I apologize for any trouble this has caused.”
Nope. NeuroNexus just out-smarted themselves with the complex numbering scheme this time around. The datasheet was wrong.
And then they sent me a $25 gift certificate to Starbucks.
This is sort of nice.
No hard feelings, NeuroNexus. (I took the lab out to coffee and it was pretty nice).
But in all seriousness, this incident left me with a few questions. Firstly, how many publications does something like this affect? I assume this adapter is not commonly used, which is one reason why I was the only person to notice it was wrong. But I am likely not the only person with the adapter, and it has been on the market in a defective form since 2011. So somebody, somewhere, probably published some messed up data. Whether the issue would compromise the findings is another question.
But on a broader level, I think the problems I had with this adapter made me reconsider the odds of simple mistakes hijacking data: these things happen. To make matters worse, there are no clear mechanisms for identifying publications in the literature that are affected once an issue is found.
So the lesson here might be: have caution with products that don’t have many end users because they implicitly won’t be scrutinized as closely. And trust your instincts if something doesn’t make sense in your data!
Adapter mapping website reflects updated map (As of February, 2016)
I still have $2.47, if anyone wants coffee.
The Corrected A32-Z32 mapping
For the handful of (1? 2?) people who might ever have this problem, here’s my corrected mapping (the channel labeled as 30 IN on the 2013 schematic (Molec input) connects to channel 27 OUT ( ZifClip output), likewise 27 IN connects to 30 OUT, 28 IN connects to 29 OUT and 29 IN connects to 28 OUT.)
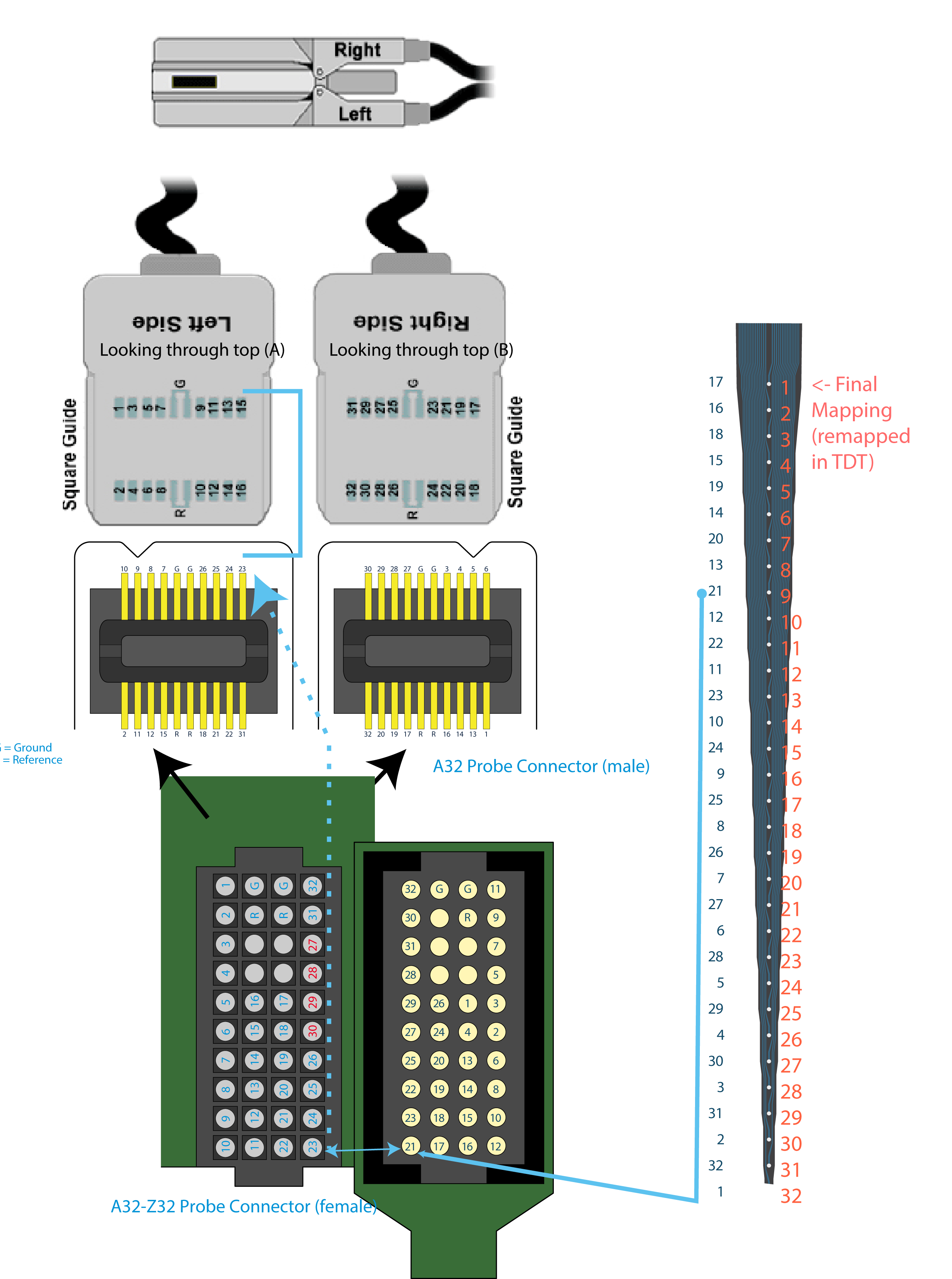
Corrected A32-Z32 mapping in red

This is great Brian. I feel your pain- we have TDT headstages and a TDT amp, but in between them is a Plexon commutator. Of course each one has it’s own channel mapping and subset of unused pins. Spent wayyy to much time making adapters so they would play nice.
Hehehe just seeing this now many years later. Hey shoot me an email some time! Hope you are well!